Purpose: The purpose of this study was to evaluate the quality of articular cartilage regeneration after arthroscopic subchondral drilling followed by postoperative intraarticular injections of autologous peripheral blood progenitor cells (PBPCs) in combination with hyaluronic acid (HA).
Methods: Five patients underwent second-look arthroscopy with chondral core biopsy. These 5 patients are part of a larger pilot study in which 180 patients with International Cartilage Repair Society grade III and IV lesions of the knee joint underwent arthroscopic subchondral drilling followed by postoperative intraarticular injections. Continuous passive motion was used on the operated knee 2 hours per day for 4 weeks. Partial weight bearing was observed for the first 6 to 8 weeks. Autologous PBPCs were harvested 1 week after surgery. One week after surgery, 8 mL of the harvested PBPCs in combination with 2 mL of HA was injected intraarticularly into the operated knee. The remaining PBPCs were divided into vials and cryopreserved. A total of 5 weekly intraarticular injections were given.
Results: Second-look arthroscopy confirmed articular cartilage regeneration, and histologic sections showed features of hyaline cartilage. Apart from the minimal discomfort of PBPC harvesting and localized pain associated with the intraarticular injections, there were no other notable adverse reactions.
Conclusions: Articular hyaline cartilage regeneration is possible with arthroscopic subchondral drilling followed by postoperative intraarticular injections of autologous PBPCs in combination with HA.
Level of Evidence: Level IV, therapeutic case series.
Adam Anz, M.D.,
Shahrin Merican, M.B.B.Ch., F.R.C.R.,
Yong-Guan Tay, M.S.Orth., F.R.C.S.(Edin),
Kunaseegaran Ragavanaidu, M.B.B.S., M.Path.,
Caroline S. Y. Jee, Ph.D.(Lond),
and David A. McGuire, M.D.
From the Kuala Lumpur Sports Medicine Centre (K-Y.S., S.M., Y-G.T.), Kuala Lumpur; Clinipath (K.R.), Klang, and Universiti Tunku Abdul Rahman (C.S.Y.J.), Kuala Lumpur, Malaysia; Wake Forest Uni- versity Baptist Medical Center (A.A.), Winston-Salem, North Carolina; and Alaska Surgery Center (D.A.M.), Anchorage, Alaska, U.S.A. The authors report no conflict of interest. Received June 21, 2010; accepted November 10, 2010. Address correspondence to Khay-Yong Saw, M.Ch.Orth., F.R.C.S.(Edin), Kuala Lumpur Sports Medicine Centre, 7th Floor, Wisma Perintis, 47 Jalan Dungun, Damansara Heights, 50490 Kuala Lumpur, Malaysia. E-mail: sportsclinic@hotmail.com © 2011 by the Arthroscopy Association of North America 0749-8063/10363/$36.00 doi:10.1016/j.arthro.2010.11.054
Chondral defects of the major weight-bearing joints currently pose an unresolved issue among orthopaedists. Marrow stimulation techniques, such as microfracture, have become the first-line treatment of small chondral defects of the knee because they are minimally invasive and have had proven results over the past 20 years.1,2 These techniques stimulate marrow repair processes that produce fibrocartilage.3,4 Recent animal studies and newer cartilage repair techniques have aimed at the regeneration of hyaline cartilage. An adjunct to marrow stimulation that achieves this goal is ideal.
Intra-articular injections of hyaluronic acid (HA) have recently proven beneficial to cartilage health and repair, illustrating the relation to increased differentiation of immature cells to chondrocytes, decreased joint inflammation, increased proteoglycan content in repair cartilage, improved histologic scores after cartilage repair, improved defect filling/incorporation after cartilage repair, and decreased postprocedural coefficients of friction.5-10 In addition, animal studies involving intra-articular injections of bone marrow– derived progenitor cells (BMPCs) and HA have documented histologic findings consistent with hyaline cartilage.8,11 BMPCs used in these scenarios have shown an innate quality allowing for the migration and integration at the sites of cartilage repair.11
In preclinical animal studies in a goat model, the first author concluded that postoperative intra-articular injections of autologous BMPCs in combination with HA after subchondral drilling resulted in improved cartilage repair.8 A clinical pilot study followed and involved standard marrow stimulation in the form of arthroscopic subchondral drilling and postoperative intra-articular injections of autologous peripheral blood progenitor cells (PBPCs) in combination with HA. Our objective was to assess whether the preclinical animal model could be replicated in the human knee joint. The purpose of this study was to evaluate the quality of resultant articular cartilage regeneration. We hypothesized that articular hyaline cartilage regeneration is possible with our novel approach.
METHODS
Patient Selection
Five patients underwent second-look arthroscopy with chondral core biopsy. These 5 patients are part of a larger pilot study in which 180 patients who presented with chondral defects of the knee joint were recruited. Postoperatively, the clinical course of these 5 patients presented an opportunity for a second-look arthroscopy. Two patients underwent contralateral knee surgery, and one patient had removal of a Tomofix plate and screw construct (Synthes, West Chester, PA), providing an opportune setting of anesthesia for second-look arthroscopy. One patient had recurrence of discomfort attributed to a prominent osteophyte and elected for a further arthroscopic procedure. The last patient had returned to football 18 months after articular cartilage repair and sustained a torn anterior cruciate ligament of the previously treated knee. He elected for arthroscopic reconstruction, which provided an opportunity for second-look arthroscopy. Informed consent after discussion of risks and benefits, as well as local ethics committee approval, was obtained before biopsy.Procedure
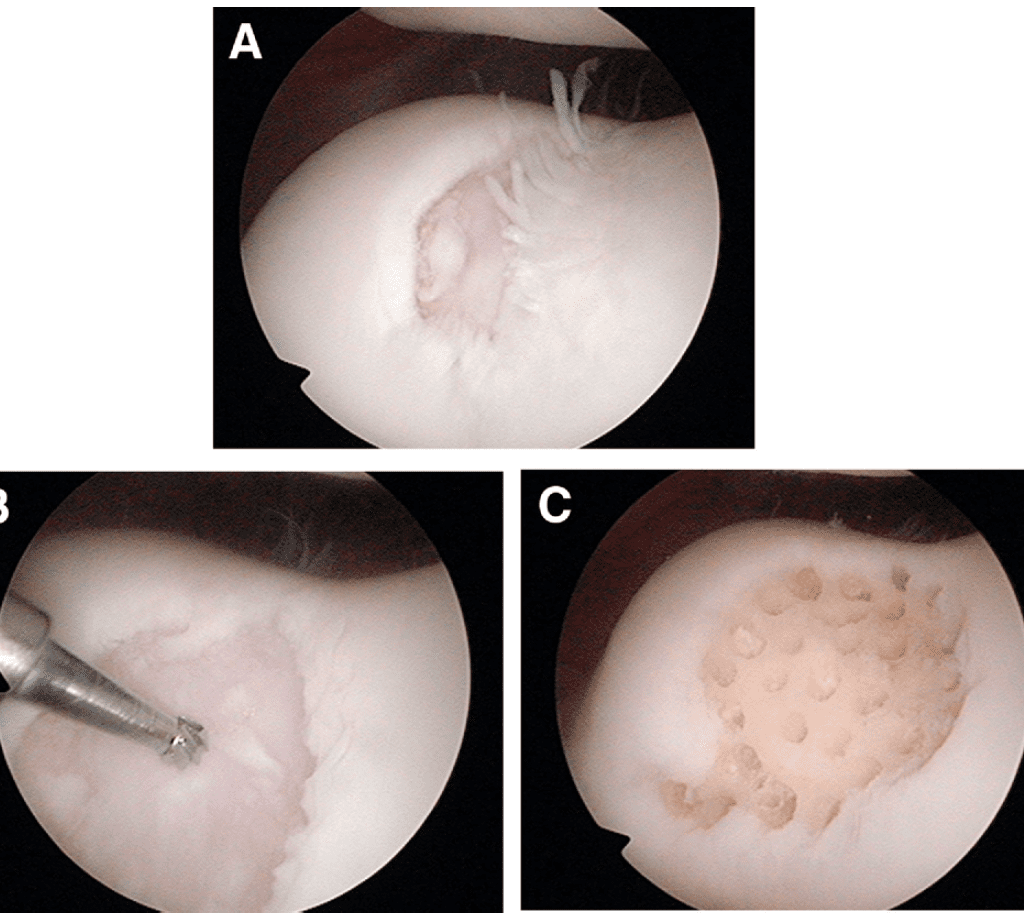
All surgical procedures were performed by a single surgeon (by the first author) involving standard arthroscopic techniques in the supine position without a tourniquet. Saline solution irrigation bags were chilled in an ice-water bath before use to minimize bleeding during the arthroscopic procedure. In our experience, we have had difficulty performing microfracture to the patella and areas of the plateau. For this reason, our preferred method is arthroscopic subchondral drilling modified from the principles established by Steadman et al13 for microfracture and Pridie14 for drilling. We begin by defining the extent of cartilage injury with a probe. A 3.5-mm full-radius shaver is used to debride loose cartilage to a stable margin; often a straight or curved arthroscopic biter is required as well. A 2-mm burr, with its guard removed, “drills” from the surface of the defect to the bone marrow, creating a conduit. The remaining area within the margin is also drilled to a depth of 5 to 10 mm. Initially, we spaced drill holes 3 to 4 mm apart. The methods have subsequently evolved so that a goal of 1 to 2 mm between drill holes is now preferred based on the results of second-look arthroscopy. It is not crucial that the subchondral drilling be performed perpendicular to the bone surface because a lesser angle of drilling capable of penetrating into the subchondral bone is sufficient. Abrasion chondroplasty up to a depth of 1 mm is performed with burring of the bony area between drill holes. The result is an extended area of bleeding bone, hence a larger surface area for the initiation of articular cartilage repair with PBPCs and HA (Fig 1). The arthroscopic portals are closed with No. 3-0 nylon suture. A mixture of 20 mL of 0.5% bupivacaine hydrochloride and epinephrine, 3 mL of 1-mg/mL morphine, and 2 mL of HA (Hyalgan; Fidia Farmaceutici, Abano Terme, Italy) is injected into the operated knee at the end of the surgical procedure.Postoperative Rehabilitation
Cold therapy is initiated immediately in the postanesthesia period and continued throughout the first month after surgery, including 1 hour 2 to 3 times per day. On the first postoperative day, continuous passive motion is used on the operated knee for a duration of 2 hours. This is continued daily for a period of 4 weeks. The range of motion is initially set at 0° to 30° and progresses as the clinical situation improves. Patients with subchondral drilling to the weight-bearing femorotibial joint are instructed on crutch-assisted partial weight bearing (15 to 20 kg) for the first 4 weeks. This progresses to full weight bearing in 6 to 8 weeks. Patients with drilling to the patellofemoral joint are allowed full weight bearing as tolerated with restrictions from weight bearing on stairs for the first 3 months after surgery. This is to avoid overloading the patellofemoral joint.
Neupogen Administration, Apheresis, and Cryopreservation
Human granulocyte colony–stimulating factor is a glycoprotein that regulates the production and release of functional neutrophils from the bone marrow. Neupogen contains recombinant granulocyte colony–stimulating factor and causes marked increases in peripheral blood neutrophil counts with a minor increase in monocytes within 24 hours. On postoperative days 4, 5, and 6, patients were given a morning dose of 300 μg of Neupogen (Filgrastim, Amgen, Thousand Oaks, CA) subcutaneously. On postoperative day 7, autologous PBPCs were collected by an automated cell separator (apheresis) by central venous access. Venous access was achieved through a femoral double-lumen catheter placed into the contralateral leg, under ultrasound guidance, performed by a consultant radiologist. Apheresis was performed by use of the Spectra Optia Apheresis Machine (Caridian BCT, Denver, CO). A fresh aliquot of 8 mL of PBPCs was separated for fresh intraarticular injection into the operated knee. The remaining PBPCs were cryopreserved in 10% dimethyl sulfoxide and divided into 4-mL cryovials for storage in liquid nitrogen at –196°C. Flow cytometry with CD34+ (hematopoietic stem cells) and CD105+ (markers for mesenchymal stem cells) was quantified. Flow cytometry was performed with a Beckman Coulter FC500 device (Beckman Coulter, Fullerton, CA).
Intra-articular Injection
On postoperative day 7, 8 mL of the fresh PBPCs is mixed with 2 mL of HA and injected into the operated knee joint under aseptic conditions in the outpatient clinic. Before this, the knee is first aspirated for hemarthrosis. At 4 subsequent weekly intervals, 8 mL (from two 4-mL cryovials) of the frozen PBPCs were obtained from the laboratory, allowed to thaw to room temperature, mixed with 2 mL of HA, and injected into the operated knee joint.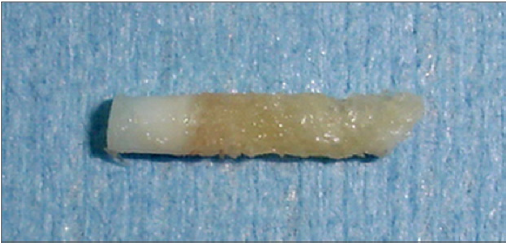
Second-Look Arthroscopy With Chondral Core Biopsy
During the second-look procedures, a chondral core biopsy specimen was procured. This was performed arthroscopically with a 5.5-mm sterilized BioCorkscrew anchor driver (Arthrex, Naples, FL). Typically, a 2-mm-diameter specimen of cartilage together with a core of bone up to 1 cm in length is obtained (Fig 2).Histology
Histologic samples were stained as follows: hematoxylin-eosin (H&E) stain was used to visualize overall morphology, safranin O was used to highlight proteoglycans, immunohistochemistry staining with anti–collagen type I mouse Ab I-8H5 stain (catalog No. CP 17; Calbiochem Merck, Darmstadt, Germany) was used to highlight collagen type I, and immunohistochemistry staining with anti–collagen type II mouse monoclonal antibody Ab 3 (clone 6B3) (catalog No. MAB8887; Millipore, Billerica, MA) was used to highlight collagen type II. Optimal dilution and predigestion with pepsin were determined by the investigator with the protocol being saved by use of software of an automated immunohistochemical slide preparation system (Ventana Benchmark; Ventana Medical Systems, Tucson, AZ).RESULTS
Second-Look Arthroscopy With Chondral Core Biopsy
Five patients underwent second-look arthroscopy, and a 2-mm chondral core biopsy specimen was procured and examined. Arthroscopically, the regenerated articular cartilage appeared smooth and had excellent integration to the surrounding native cartilage without any delamination or hypertrophy. The exception was case 2, in which the drill holes over the lateral patellofemoral joint were too far apart with resultant tufts of cartilage seen between areas devoid of regenerated cartilage (Fig 3).Histology
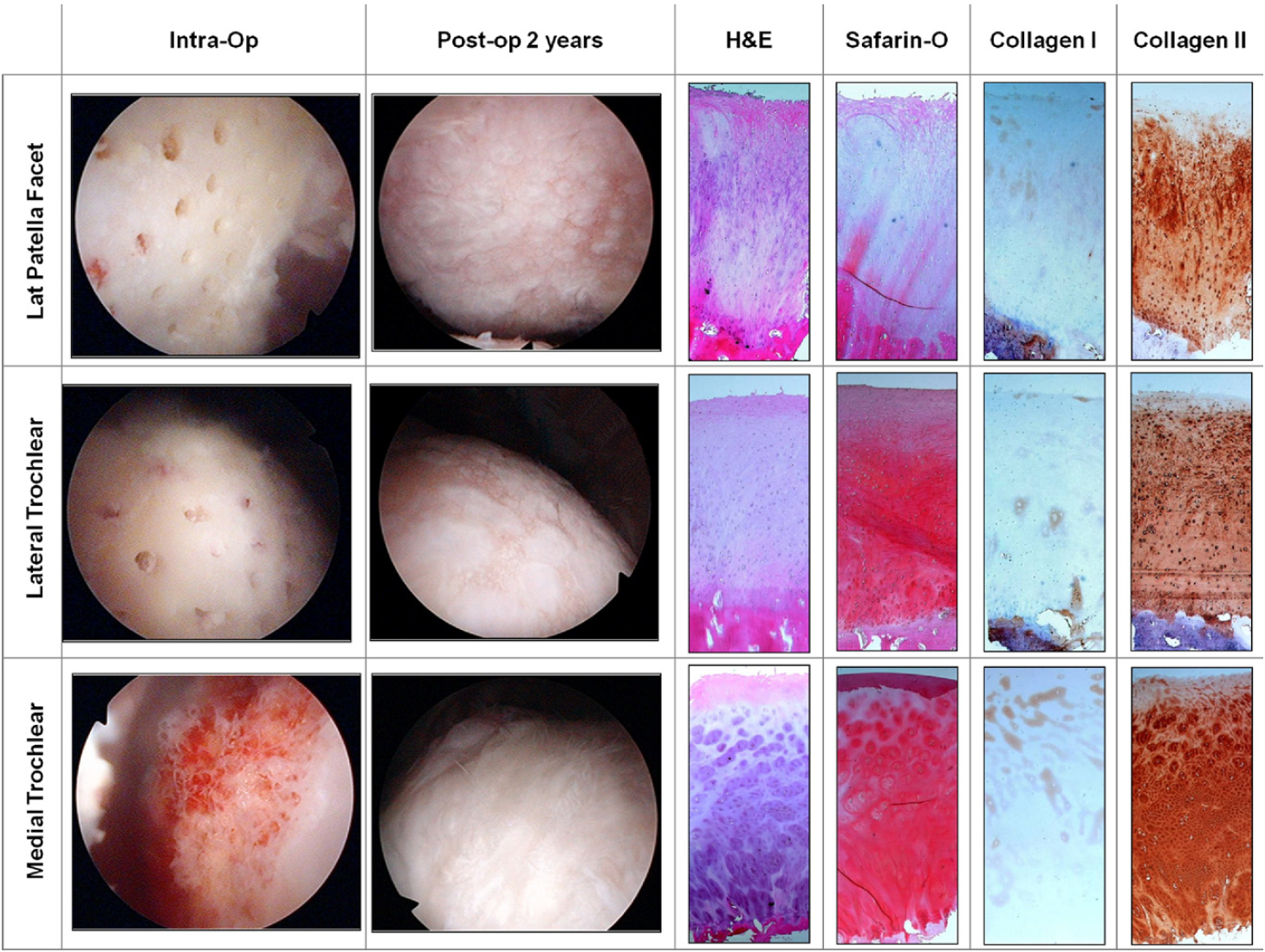
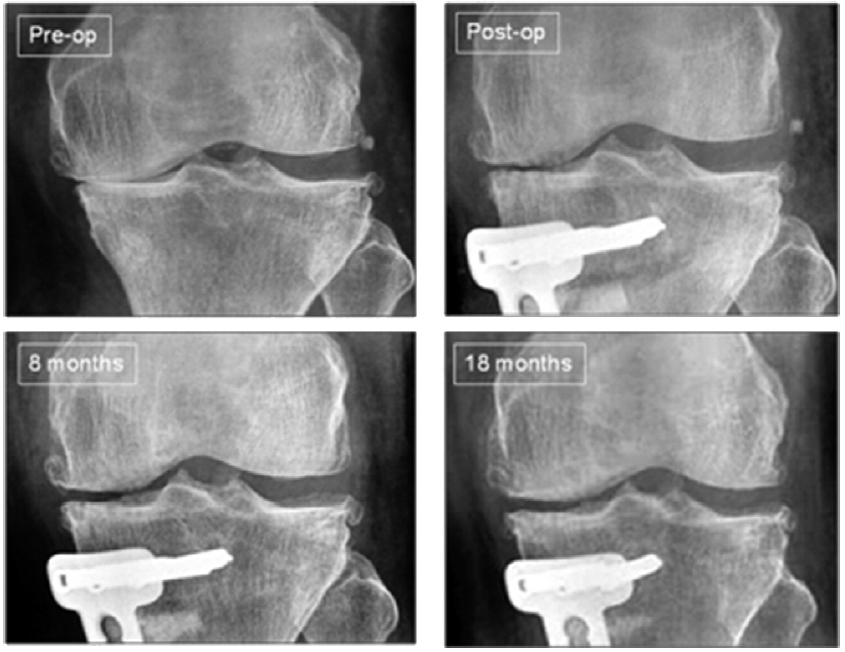
Cases 1 and 2 with gross grade IV kissing lesions are presented with multiple biopsy specimens and histologic analyses after second-look arthroscopy. Cases 3, 4, and 5 are patients with smaller isolated lesions.Case 1: Biopsy was performed 22 months after the initial surgery in a 49-year-old woman with a varus deformity who underwent debridement, subchondral drilling, and an open wedge high tibial osteotomy with Tomofix fixation (Fig 4). Approximately 80% of the weight-bearing medial compartment had grade III and IV lesions. Weight-bearing radiographs at 8 and 18 months showed reappearance of the medial femorotibial joint space. Second-look arthroscopy of the regenerated cartilage showed a stable, smooth surface with no delamination. On probing, the regenerated cartilage had the same consistency as the surrounding normal cartilage. The second-look images and biopsy specimens are included in Fig 5.Immunohistochemistry staining was performed to assess the collagen type I and type II content of the biopsy specimens. Specimens from the medial femoral condyle and medial tibial plateau showed the presence of collagen type I confined to the superficial layer. Collagen type II was present throughout the deeper layers. These are features of hyaline cartilage as opposed to fibrocartilage.
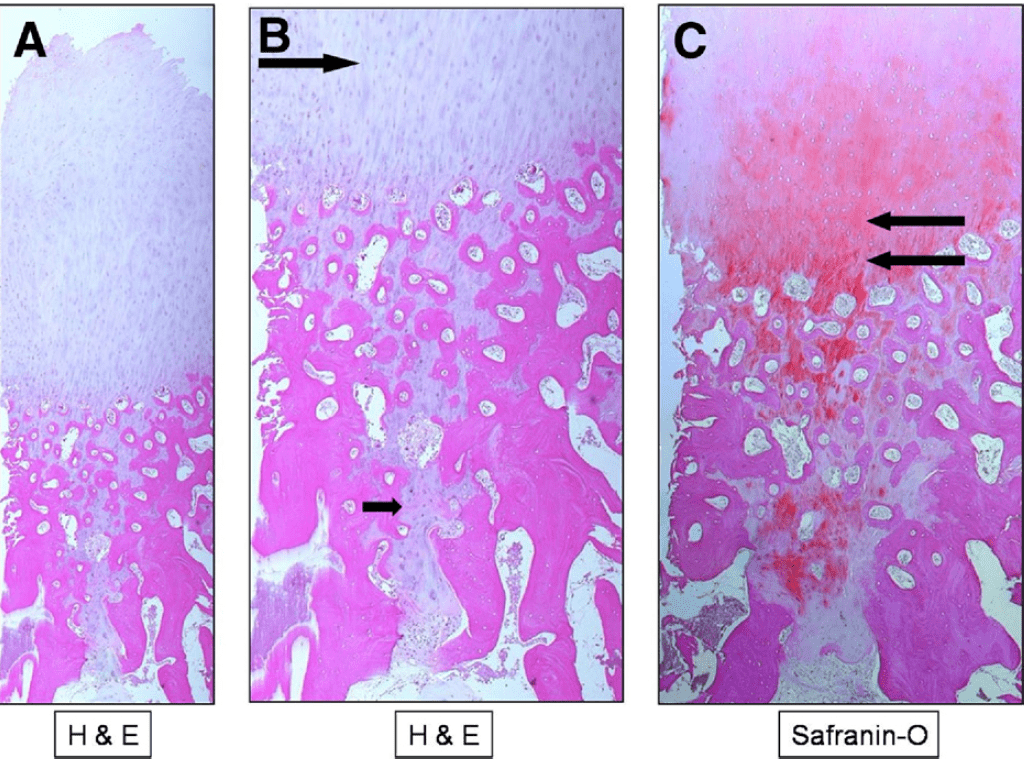
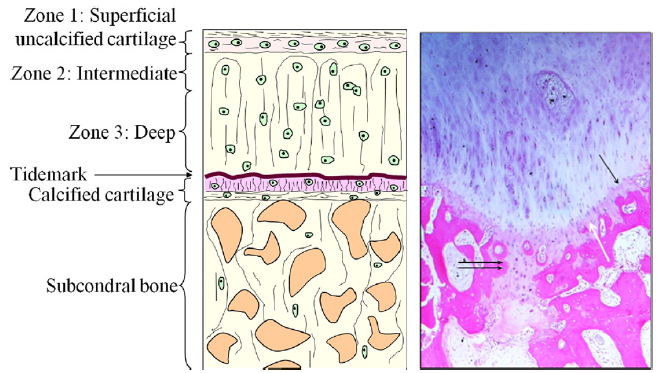
One full-thickness biopsy specimen from the medial tibial plateau captured a drill hole and adjacent bone (Fig 6). This sample shows full-thickness regenerated articular cartilage with a fairly smooth articular surface, subchondral bone, and marrow space. A streaming, linear pattern of chondrocytes is seen arising from the subchondral bone region from the area of previous subchondral drilling. Incidental findings of cartilage clusters with early ossification are also seen within the tract. The chondrocytes are involved in ongoing remodeling and are present beneath the calcified cartilage layer. A biopsy specimen from the medial femoral condyle of the patient also showed the presence of a previous drill hole lined with chondrocytes. This sample illustrated an area of regeneration undergoing ossification, showing re-establishment of the calcified cartilage layer and the tidemark (Fig 7). The tidemark is seen as an undulating basophilic line on routine H&E staining and is the point at which the articular cartilage becomes calcified. The subchondral drill defect area is replaced by the presence of chondrocytes with varying degrees of maturation surrounded by a ground substance matrix. In addition, ossification is evident with new trabecular bone formation. Ossification changes were not seen over the new hyaline cartilage formation zone.
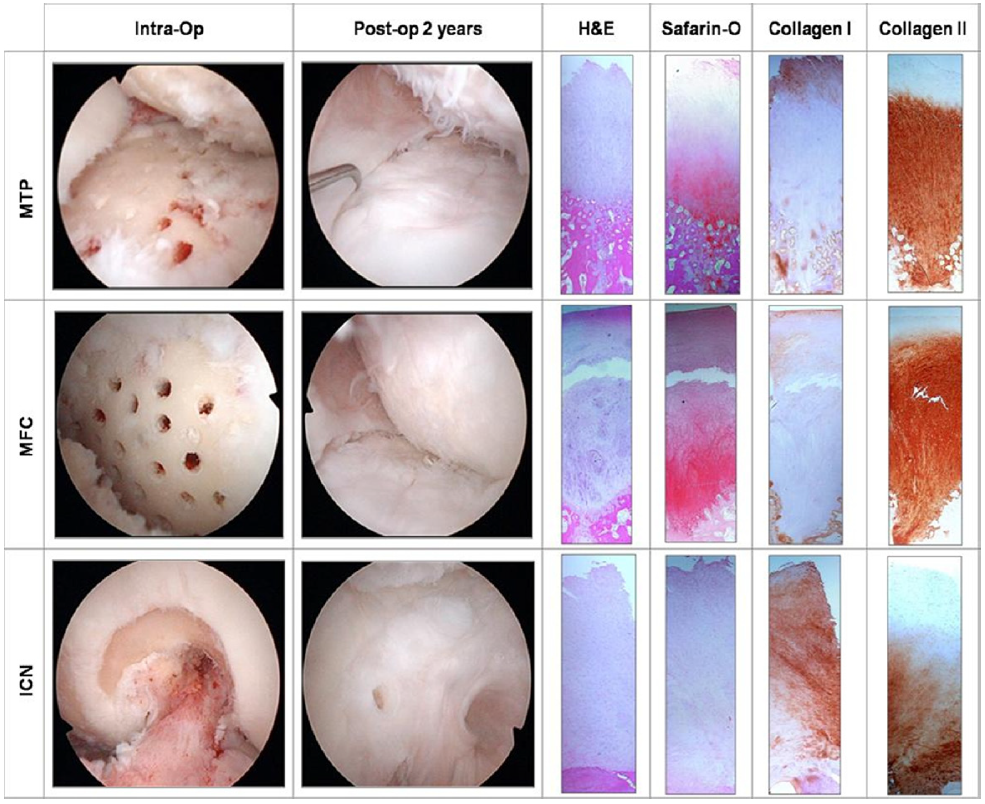
The intercondylar notch sample was an area of previous roof-plasty and notchplasty with abrasion chondroplasty. This represents an area that is non– weight bearing. Histologic biopsy examination here showed a mixed tissue with fibrocartilage and hyalinelike cartilage: the predominance of collagen type I, less collagen type II, and chondrocytes arranged in a more disorganized pattern. We believe that the failure of the chondrocytes to exhibit a linear streaming pattern, as seen from the medial tibial plateau and medial femoral condyle biopsy specimens, is due to the absence of a weight-bearing force on the intercondylar notch (Fig 5).
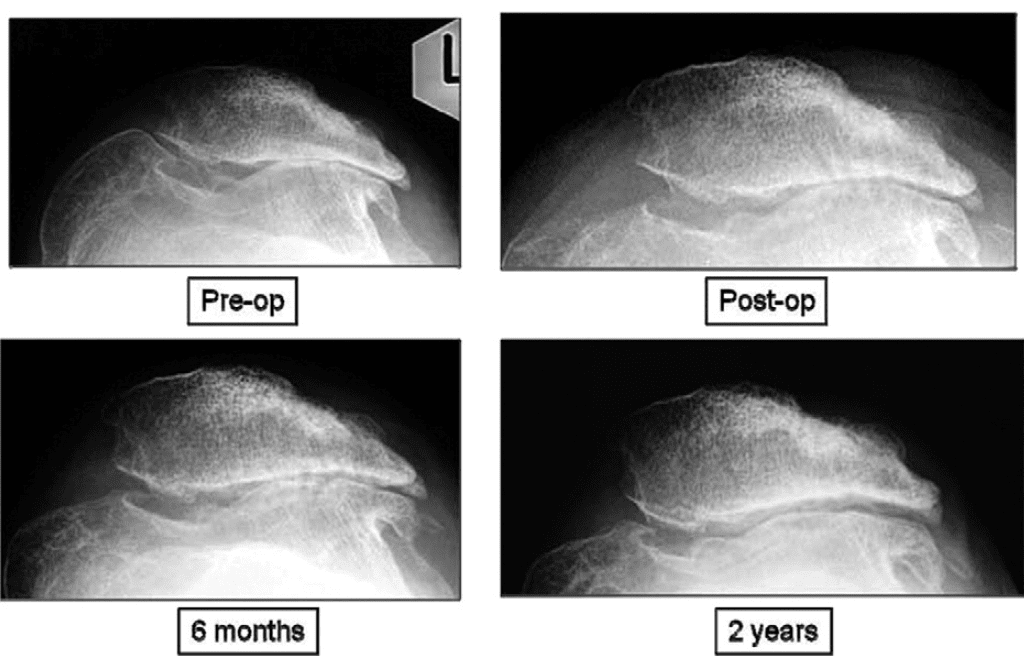
Case 2: A chondral core biopsy specimen was taken 26 months after surgery in a 34-year-old woman with previous multiple open surgeries for recurrent dislocation of the patella as an adolescent. She underwent arthroscopic debridement, lateral patella release, and subchondral drilling. There were grade III and IV lesions over the entire patellofemoral joint. Preoperative merchant-view radiographs showed severe patellofemoral osteoarthritis, a large medial trochlear osteophyte, and absence of the lateral patellofemoral joint space (Fig 8). During arthroscopic surgery, the medial trochlear osteophyte was burred and subchondral drilling was performed on the entire patellofemoral joint. An immediate postoperative radiograph showed evidence of subchondral drilling.
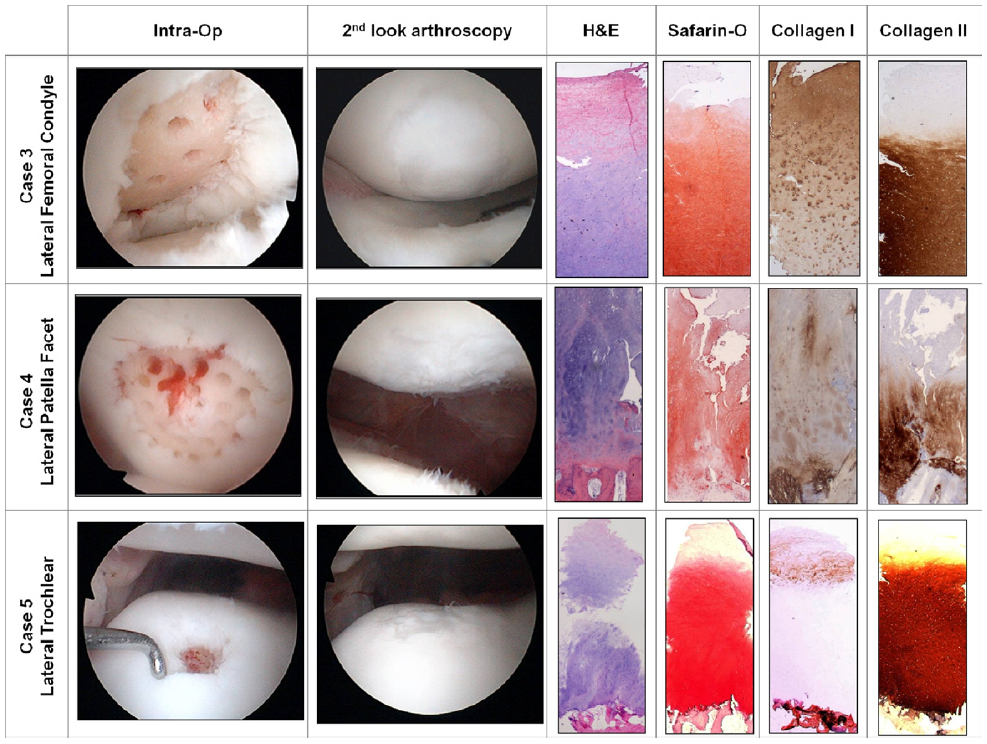
Case 3: A biopsy specimen was obtained 1 year after surgery from a 52-year-old woman with an isolated grade IV lesion of the lateral femoral condyle measuring 2 × 1 cm.
Case 4: A biopsy specimen was obtained 10 months after surgery in a 43-year-old woman with lateral patellar maltracking. There was a grades III and IV defect measuring 2.5 × 3.5 cm over the lateral patella facet. Lateral patella release was performed in addition to subchondral drilling.
Case 5: Biopsy was performed at 18 months after surgery in a 19-year-old man with lateral patellar maltracking and a lateral trochlear grade IV lesion measuring 0.8 cm in diameter. Subchondral drilling was performed followed by lateral patellar release (Fig 9).
Chondral core biopsy specimens in cases 2, 3, 4, and 5 with histologic staining with H&E showed columnar morphology of cells with a pale blue ground substance. Safranin O showed intense orange/red staining of the newly regenerated cartilage zone throughout the regenerated cartilage layer with a propensity toward the deeper areas of cartilage above the subchondral bone. The matrix also showed a predominance of collagen type II deposits, whereas collagen type I was minimal and located mostly over the superficial regions of the articular surface. These compositional results are features of hyaline as opposed to fibrocartilage.5,8,11
DISCUSSION
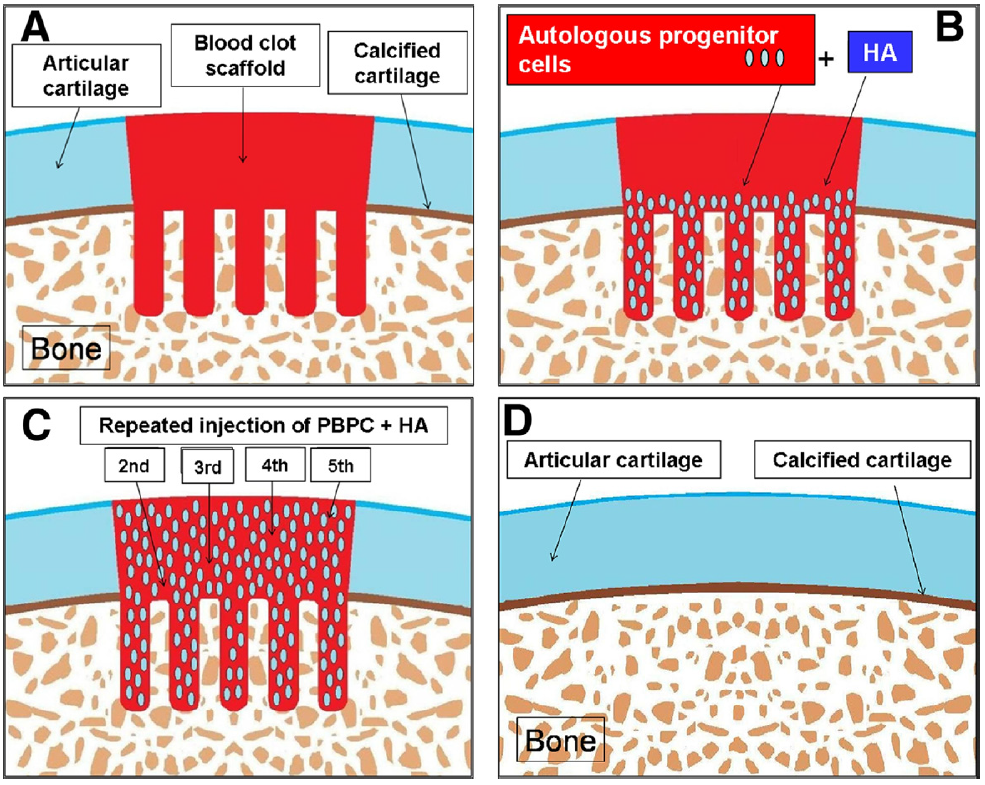
It was possible to replicate our preclinical animal model8 in the human knee joint using postoperative intraarticular injections of autologous PBPCs in combination with HA after arthroscopic subchondral drilling. This method uses marrow stimulation to create an autologous scaffold that is subsequently seeded with postoperative intraarticular injections of autologous PBPCs and in situ progenitor cells from the marrow (Fig 10).Histologic results after microfracture and autologous chondrocyte implantation (ACI) in humans are limited because of ethics and the specifics of obtaining regenerative tissue samples. Peterson et al.15 presented the earliest histologic results concerning ACI. They obtained biopsy specimens from 37 of 101 patients with 2 to 9 years’ follow-up. They reported on 21 biopsy specimens from patients with isolated femoral condyle lesions, osteochondritis dissecans, and femoral lesions with anterior cruciate ligament repair. They reported that 17 cases had homogeneous matrix with low cellularity, which they termed hyaline-like. Collagen type II staining for these samples was positive. Of these 21 biopsy specimens, 4 represented irregular patterns with high cellularity and negative collagen type II staining.These were determined to represent fibrocartilage. Two of their specimens had a mixed appearance. They did not report results on the remaining 12 biopsy specimens from patients who had either patellar lesions with realignment or multiple lesions.15
Further recent studies have had more mixed results when compared with those of Peterson et al.15 Hollander et al.16 used quantitative assays for collagen type I, collagen type II, and proteoglycan to evaluate 23 patients treated with a refined method of ACI. Using guidelines from the Histological Endpoint Committee of the ICRS, they reported on 10 patients with hyaline cartilage, 3 patients with mixed tissue, and 10 patients with fibrocartilage. Bartlett et al.17 reported on 91 patients who underwent 2 types of ACI, 1 with a collagen cover and 1 with a collagen matrix. In total, 25 patients underwent biopsy, and the authors found 7 with hyaline-like tissue, 3 with mixed tissue, and 15 with fibrocartilage. According to the 5-year results of Knutsen et al.18 on 80 patients randomized to microfracture versus ACI, 67 patients underwent biopsy; 10 of 67 exhibited predominantly hyaline tissue, and 29 of 67 exhibited fibrocartilage. No difference was found between the microfracture and ACI groups when comparing the frequency of hyaline and fibrocartilage.
All of our biopsy specimens showed histologic features of hyaline cartilage with anti–collagen type I stain (used to highlight collagen type I), anti–collagen type II stain (used to highlight collagen type II), and safranin O stain (used to highlight proteoglycans), with the exception of 1 histologic sample showing mixed cartilage. This biopsy specimen was from an area of abrasion notchplasty, which represented a non–weight-bearing region. Comparison of biopsy specimens from this non–weight-bearing area to those from a weight-bearing area in the same patient has led us to theorize that early partial weight bearing is essential for the regeneration and alignment of collagen type II. In addition, all of our areas of repair produced smooth cartilage with the exception of case 2, in which the drilling over the lateral patellofemoral joint was spaced at 3 to 5 mm apart, resulting in tufts of cartilage between areas devoid of regenerated cartilage (Fig 3). As a result, we have refined our techniques so that a goal of 1 to 2 mm between drill holes is sought. Abrasion chondroplasty up to a depth of 1 mm is also performed between the drill holes (Figs 11 and 12).
Treatment involving progenitor cells stems from the hematology/oncology profession: Given the potential morbidity associated with iliac crest harvest, cell collection for bone marrow repopulation now involves mobilization of multipotential progenitor cells through hormonal stimulation and collection through a peripheral automated cell separator machine. This process, commonly referred to as apheresis, has potential for increased magnitude of harvest. Studies involving review of healthy donors have shown this to be a safe and effective procedure for the production and harvest of cells.19,20 Recent study into the properties of these PBPCs has shown that they are similar to embryonal stem cells in that they express transcription factors specific to pluripotential cells, have proliferative potential, have the ability to differentiate into a multitude of cell types, and are more immature than BMPCs.19 In addition, when injected subcutaneously into mice, these cells were found to migrate to multiple organs and integrate and function as the surrounding cells.19 In addition to implementing evidence from recent animal studies, we have sought to make use of clinical evidence regarding the potential and safety of PBPCs, preferring to use PBPCs as opposed to BMPCs because of the ease of harvest, decreased harvest-site morbidity, and increased potential with these cells.19-21
Table 1 shows the PBPC count of 20 recent consecutive patients after refinement of our processing methods, showing data of fresh and frozen samples with white blood cell count, CD34+ and CD105+ counts, and cell viability in relation to the number of Neupogen injections administered.
Fresh cells are used preferably for the first injection because of a mean viability of 99% compared with frozen cells, which have a mean viability of 87%. It should be noted that 8 mL of PBPCs injected into the operated knee has a mean of 20 million CD105+ cells. Historically, the cell marker CD34+ (hematopoietic stem cells) has been used to identify functional cells for bone marrow transplant. We have begun to draw interest in CD105+ cells, because this is the marker for mesenchymal stem cells. Five weekly injections are based on the HA protocol for osteoarthritis, as well as the suggestion from preclinical animal studies involving BMPCs that an increased number of intraarticular injections is more efficacious.8
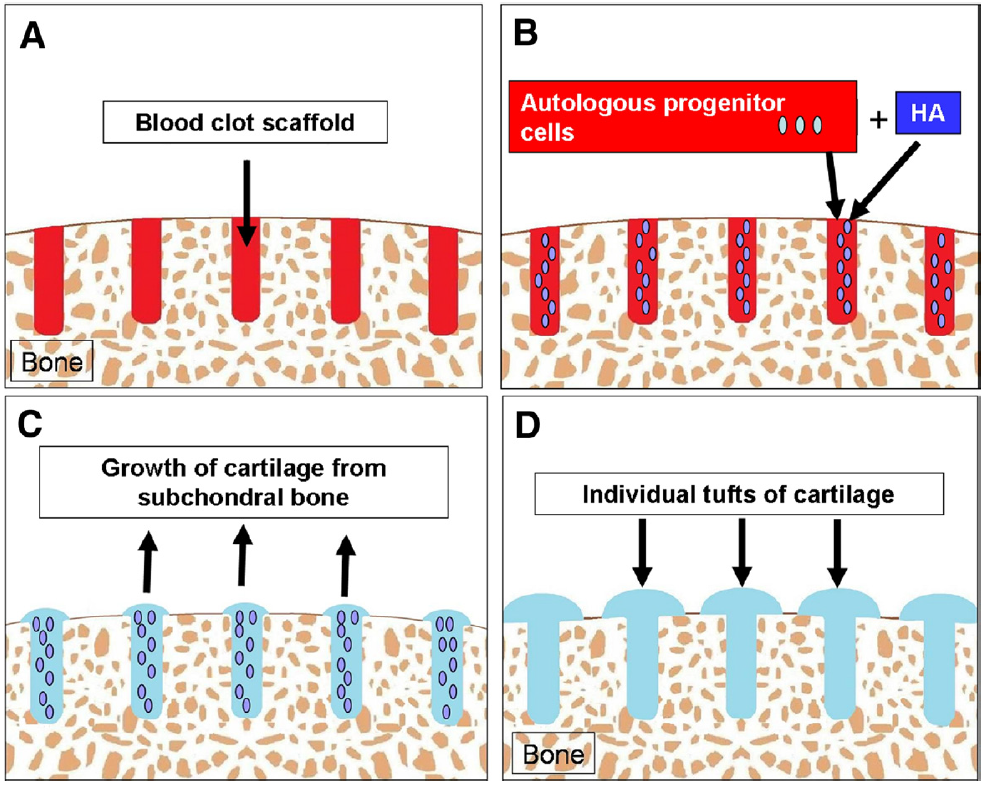
Our theory is that providing a high percentage of immature multipotential progenitor cells into the right environment allows these cells to populate areas of subchondral drilling and regenerate hyaline cartilage. Our histologic findings of chondrocytes below the calcified cartilage layer at a subchondral drill hole (Figs 6 and 7) and the porcine model of Lee et al.11 illustrating mesenchymal stem cells at the base of newly formed cartilage support the idea that injected progenitor cells are attracted to the site of marrow injury, proliferate into chondrocytes, and regenerate hyaline cartilage from the subchondral base.
We theorize that the addition of matrix substance, in the form of HA and passive stimulating kinetic movement of the involved joint (continuous passive motion), provides chemical and cellular signals for regeneration and aids synovial movement. Partial to full weight bearing in the early phase of rehabilitation provides the essential environment to assist in the remodeling of the collagen fibrils to align along the axis of weight transmission.
The limitations of this study are lack of a control group, small number of patients, and short-term follow-up. The analysis of histologic results was subjective and not based on comparison with a control group. This offers some observational bias. A randomized controlled trial comparing a group with PBPC and HA injections with a group with HA injections alone is currently under way, being supervised by the first author.
CONCLUSIONS
Articular hyaline cartilage regeneration is possible with arthroscopic subchondral drilling followed by postoperative intraarticular injections of autologous PBPCs in combination with HA.
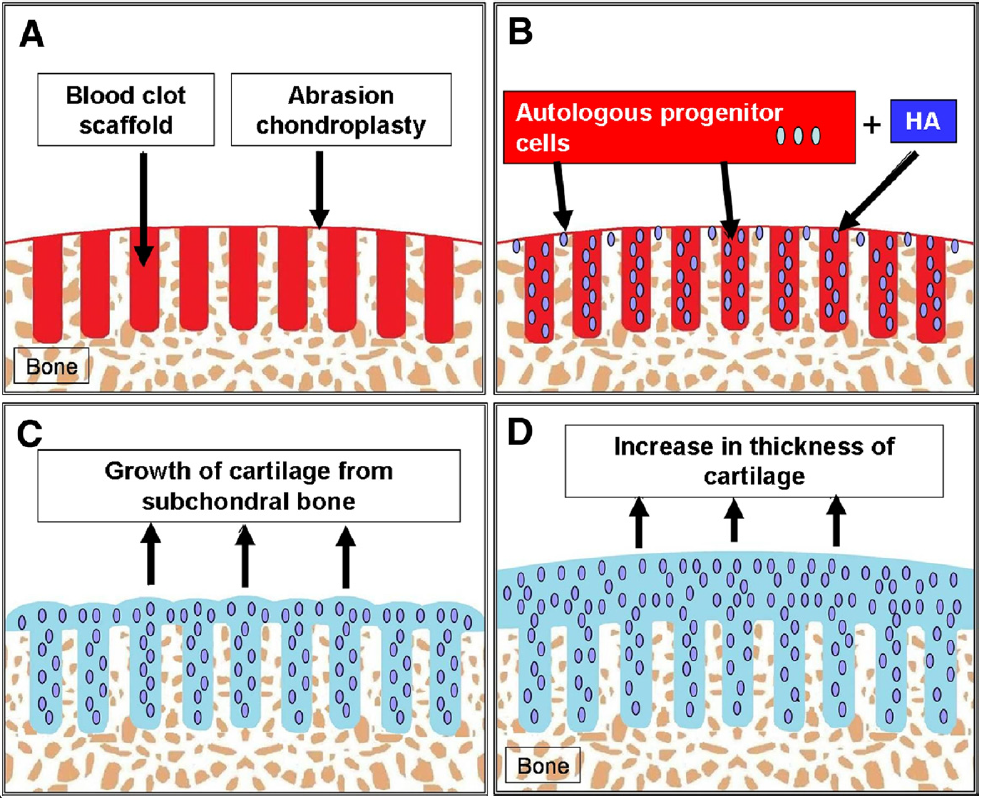
FIGURE 12. Chondrogenesis: Uncontained lesion—ideal drilling. (A) Ideally placed subchondral drilling (1 to 2 mm apart) and abrasion
chondroplasty between the drill holes increase the available bony areas for the homing of the PBPCs. (B) Injected PBPCs and HA have a
larger surface area of raw bone providing homing signals for the recruitment of the PBPCs. (C) Individual tufts of cartilage arise from the
subchondral bone and coalesce to cover the bony defect. (D) Maturation will result in an increase in thickness of the regenerated cartilage
covering the entire defect.
| White Blood Cell Count 103/μL | CD34+ (106) Cells per 4-mL Vial | CD105+ (106) Cells per 4-mL Vial | Viability % | ||||
|---|---|---|---|---|---|---|---|
| Fresh | Frozen | Fresh | Frozen | Fresh | Frozen | ||
| Case 1 | 32.00 | 6.86 | 4.58 | 7.42 | 8.14 | 99.30 | 79.90 |
| Case 2 | 24.40 | 1.04 | 0.66 | 9.98 | 14.41 | 99.30 | 89.65 |
| Case 3 | 30.00 | 0.33 | 0.23 | 3.32 | 5.08 | 99.10 | 89.99 |
| Case 4 | 35.60 | 1.03 | 0.74 | 15.44 | 20.33 | 99.01 | 91.97 |
| Case 5 | 14.80 | 3.88 | 2.86 | 2.69 | 8.87 | 99.40 | 87.90 |
| Case 6 | 33.50 | 0.44 | 0.37 | 8.95 | 7.04 | 99.60 | 95.30 |
| Case 7 | 33.50 | 3.28 | 2.33 | 5.41 | 7.75 | 99.00 | 82.90 |
| Case 8 | 34.80 | 2.02 | 1.34 | 13.60 | 14.01 | 99.50 | 94.04 |
| Case 9 | 23.00 | 2.53 | 1.69 | 7.01 | 13.77 | 99.50 | 88.10 |
| Case 10 | 37.10 | 2.42 | 1.32 | 3.37 | 5.72 | 98.90 | 88.10 |
| Case 11 | 26.50 | 1.55 | 1.07 | 4.54 | 6.98 | 99.00 | 84.80 |
| Case 12 | 52.10 | 2.76 | 2.17 | 8.89 | 4.56 | 99.40 | 90.30 |
| Case 13 | 38.80 | 3.15 | 1.82 | 15.50 | 11.62 | 99.30 | 79.30 |
| Case 14 | 32.10 | 0.86 | 0.75 | 12.79 | 6.35 | 98.90 | 91.30 |
| Case 15 | 33.00 | 5.71 | 3.14 | 6.30 | 8.53 | 99.10 | 78.40 |
| Case 16 | 20.00 | 1.02 | 0.62 | 14.80 | 15.40 | 97.80 | 88.40 |
| Case 17 | 37.50 | 3.19 | 2.08 | 4.15 | 1.66 | 99.30 | 82.20 |
| Case 18 | 26.90 | 2.78 | 1.90 | 1.43 | 4.44 | 99.30 | 82.30 |
| Case 19 | 31.00 | 1.55 | 1.01 | 23.45 | 31.15 | 99.10 | 89.10 |
| Case 20 | 44.60 | 1.16 | 0.67 | 16.62 | 19.40 | 98.50 | 91.80 |
| Mean | 32.06 | 2.38 | 1.57 | 9.28 | 10.76 | 99.12 | 87.29 |
REFERENCES
- Magnussen RA, Dunn WR, Carey JL, Spindler KP. Treatment of focal articular cartilage defects in the knee: A systematic review. Clin Orthop Relat Res 2008;466:952-962.
- McGuire DA, Carter TR, Shelton WR. Complex knee reconstruction: Osteotomies, ligament reconstruction, transplants, and cartilage treatment options. Arthroscopy 2002;18:90-103 (Suppl 2).
- Menche DS, Frenkel SR, Blair B, et al. A comparison of abrasion burr arthroplasty and subchondral drilling in the treatment of full-thickness cartilage lesions in the rabbit. Arthroscopy 1996;12:280-286.
- Steinwachs MR, Guggi T, Kreuz PC. Marrow stimulation techniques. Injury 2008;39:S26-S31 (Suppl 1).
- Kang SW, Bada LP, Kang CS, et al. Cartilage regeneration with microfracture and hyaluronic acid. Biotechnol Lett 2008;30:435-439.
- Kaplan LD, Lu Y, Snitzer J, et al. The effect of early hyaluronic acid delivery on the development of an acute articular cartilage lesion in a sheep model. Am J Sports Med 2009;37:2323-2327.
- Lane J, Healey R, Amiel D. Changes in condylar coefficient of friction after osteochondral graft transplantation and modulation with hyaluronan. Arthroscopy 2009;25:1401-1407.
- Saw KY, Hussin P, Loke SC, et al. Articular cartilage regeneration with autologous marrow aspirate and hyaluronic acid: An experimental study in a goat model. Arthroscopy 2009;25:1391-1400.
- Strauss E, Schachter A, Frenkel S, Rosen J. The efficacy of intraarticular hyaluronan injection after the microfracture technique for the treatment of articular cartilage lesions. Am J Sports Med 2009;37:720-726.
- Tytherleigh-Strong G, Hurtig M, Miniaci A. Intraarticular hyaluronan following autogenous osteochondral grafting of the knee. Arthroscopy 2005;21:999-1005.
- Lee KB, Hui JH, Song IC, Ardany L, Lee EH. Injectable mesenchymal stem cell therapy for large cartilage defects—A porcine model. Stem Cells 2007;25:2964-2971.
- Brittberg M, Peterson L. Introduction of an articular cartilage classification. International Cartilage Repair Society Newsletter 1. 1998:5-8.
- Steadman JR, Rodkey WG, Briggs KK, Rodrigo JJ. The microfracture technique in the management of complete cartilage defects in the knee joint. Orthopade 1999;28:26-32.
- Pridie KH. A method of resurfacing osteoarthritic knee joints. J Bone Joint Surg Br 1959;41:618-619.
- Peterson L, Minas T, Brittberg M, Nilsson A, Sjogren-Jansson E, Lindahl A. Two- to 9-year outcome after autologous chondrocyte transplantation of the knee. Clin Orthop Relat Res 2000:212-234.
- Hollander AP, Dickinson SC, Sims TJ, et al. Maturation of tissue engineered cartilage implanted in injured and osteoarthritic human knees. Tissue Eng 2006;12:1787-1798.
- Bartlett W, Skinner JA, Gooding CR, et al. Autologous chondrocyte implantation versus matrix-induced autologous chondrocyte implantation for osteochondral defects of the knee: A prospective, randomised study. J Bone Joint Surg Br 2005;87:640-645.
- Knutsen G, Drogset JO, Engebretsen L, et al. A randomized trial comparing autologous chondrocyte implantation with microfracture. Findings at five years. J Bone Joint Surg Am 2007;89:2105-2112.
- Cesselli D, Beltrami AP, Rigo S, et al. Multipotent progenitor cells are present in human peripheral blood. Circ Res 2009;104:1225-1234.
- Holig K, Kramer M, Kroschinsky F, et al. Safety and efficacy of hematopoietic stem cell collection from mobilized peripheral blood in unrelated volunteers: 12 Years of single-center experience in 3928 donors. Blood 2009;114:3757-3763.
- Ordemann R, Holig K, Wagner K, et al. Acceptance and feasibility of peripheral stem cell mobilisation compared to bone marrow collection from healthy unrelated donors. Bone Marrow Transplant 1998;21:S25-S28 (Suppl 3).
For additional information on ACL knee injuries, or to learn more about what is involved during ACL reconstruction surgery, please contact the office of orthopedic knee surgeon, Dr. Adam Anz, serving the greater Pensacola, Gulf Breeze, and Gulf Coast communities.



